

Journal of Medical Sciences and Health
DOI: 10.46347/jmsh.v12.i1.25.203
Year: 2026, Volume: 12, Issue: 1, Pages: 82-90
Original Article
Sharavi Budanur 1, A S Sanjana 2, Tarun Kumar D Jain 1
1 Junior Resident, Department of Dermatology, Venereology and Leprosy, BGS GIMS Hospital, Karnataka, India.
2 Professor and Head of Department, Department of Dermatology, Venereology and Leprosy, BGS GIMS Hospital, Karnataka, India.
Address for correspondence: Tarun Kumar D Jain, Junior Resident, Department of Dermatology, Venereology and Leprosy, BGS GIMS Hospital, Karnataka, India.
E-mail: [email protected]
Received Date:27 May 2025, Accepted Date:13 March 2026, Published Date:06 May 2026
Background: Acne vulgaris is a prevalent skin disorder with significant physical and psychological implications, particularly among adolescents and young adults. Recent research suggests a potential link between acne severity and metabolic parameters, including metabolic syndrome and insulin resistance.
Objectives: This study aimed to investigate the relationship between acne severity and metabolic parameters in post-adolescent male patients. The objectives were to determine the prevalence of metabolic syndrome and insulin resistance among acne patients, assess acne severity using a global grading system, and describe the demographic profile of the study participants.
Methods: A hospital-based cross-sectional observational study was conducted among male patients aged 20-44 years presenting to the Department of Dermatology at BGS GIMS, Bangalore, from August 2022 to July 2023. A total of 126 participants with acne vulgaris were purposively selected. Demographic details were collected using pre-structured proforma. Metabolic syndrome was diagnosed based on NCEP-III criteria, while insulin resistance was defined as a HOMA-IR value ≥2.5.
Results: The study reported a prevalence of metabolic syndrome of 18.3%, with higher incidence observed among participants aged 26-30 years. Insulin resistance was observed in 32.5% of the participants. The distribution of raised HOMA-IR values varied across age groups. Among participants with raised HOMA-IR values, 36.6% belonged to grade II acne severity, while 29.3% were categorized as grade III. Among those with metabolic syndrome, 56.5% had acne since ≤1 year, indicating an early onset of metabolic disturbances in this population. Conclusions: This study highlights the intricate interplay between metabolic parameters and acne severity in postadolescent males.
Acne is the most common skin disorder among adolescents, with a prevalence of 35%-90%. It peaks between the ages of 14 and the beginning of the third decade, but it can last into adulthood or develop de novo (20% men and 35% women). Acne is caused by the interaction of four factors: follicular hyperkeratinisation with follicular clogging, increased sebum production, the release of inflammatory mediators, and Propionibacterium acnes follicle colonisation[1-7]. Acne vulgaris is a multifactorial pilosebaceous unit disease. Clinically picture ranges from mild comedonal acne to severe systemic illness. Acne has a clear economic and psychosocial impact, sometimes causing self-
consciousness and social isolation in individuals who suffer from it. Scarring, depigmentation, sadness, anxiety, and low self-esteem are among the side effects of acne. Acne lesions are more commonly found on the face, chest, or upper back. The lesions may be non-inflammatory closed comedones (i.e. also called whiteheads); open comedones (i.e. also called blackheads); or inflammatory papules, nodules, pustules, and cysts. Acne severity is graded as mild, moderate, or severe based on the size and kind of lesions[8]. The role of insulin in acne is that it stimulates the synthesis of androgens, which are important in the development of excess sebum, hyperkeratinisation, and sebaceous gland proliferation. As a result, insulin may play a role in the aetiology and severity of acne. Acne is caused by the role of lipid metabolism and hormonal activity in sebocyte development. IGF-1 (insulin-like growth factor-1) has also been demonstrated to promote increased sebum production and acne on its own. A prior study found higher IGF-1 levels in acne patients, possibly indicating a role for insulin and growth hormone levels[9-11]. In 2009, the American Heart Association (AHA) and the National Heart, Lung, and Blood Institute (NHLBI) reached an agreement that included central obesity as one of the
criteria. It also suggested including ethnicity-specific waist circumference (separately for males and females)[12]. Other factors include high triglyceride levels, low high-density lipoproteins, high blood pressure, and high fasting blood sugar. Metabolic Syndrome (MetS) is defined as the existence of any three of the five characteristics in an individual. MetS pathogenesis is tightly linked to insulin resistance. Muscles, fat, and other cells become insensitive to insulin levels in the bloodstream and are unable to absorb glucose[13]. Insulin resistance is exacerbated by central obesity and adipose tissue build-up. MetS causes a variety of hormonal abnormalities due to the accumulation of extra adipose tissue and insulin resistance[14]. Inflammatory markers such as TNF-, IL 17, IL-23, and oxidative stress have been linked to the pathophysiology of MetS and acne[15]. With this background in mind, we tried to determine the association of acne in post adolescent male patients with metabolic syndrome and insulin resistance and to also assess acne grade with a global acne grading system.
Among the patients presenting to the Department of Dermatology, venereology, leprosy at a tertiary care centre-
To determine the association of acne in post-adolescent male patients with metabolic syndrome.
To determine the association of acne in post-adolescent male patients with insulin resistance.
To assess acne grade with a global acne grading system in the patients having metabolic syndrome and/or insulin resistance.
To describe the demographic profile of post-adolescent male patients with acne.
Study design: A hospital based cross-sectional observational study was conducted among patients presenting to the Department of Dermatology, venerology, leprosy, from August 2022 to July 2023.
Study duration: The study was conducted over a period of 12 months from August 2022 to July 2023.
Study participants: Male patients having acne vulgaris in the age group of 20-44 years presenting to the Department of Dermatology.
Sampling technique: Male patients having acne vulgaris in the age group of 20-44 years presenting to the Department of Dermatology, was selected purposively in the study.
Inclusion criteria
Male patients having acne vulgaris in the age group of 20-44 years.
Exclusion Criteria
1. Patients on treatment for acne vulgaris
2. Patients on lipid lowering drugs and hypoglycaemic drugs
3. Patients on antiepileptics, steroids, anti-tubercular drugs, antipsychotics
Study procedure: After obtaining permission from the Research monitoring committee and Institutional Ethics Committee (IEC), the study was conducted in Dermatology Department. Patients fulfilling inclusion and exclusion criteria were enrolled in the study. Study procedures were explained to the participant and written informed consent was taken after providing patient information sheet.
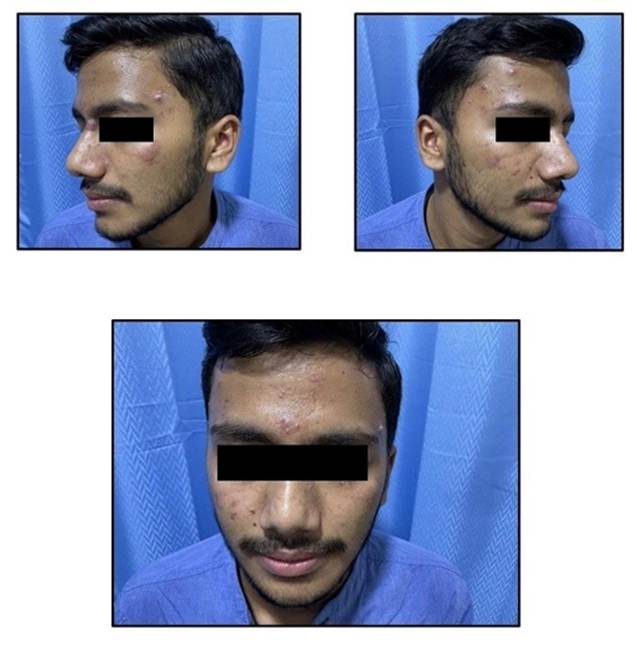
A pre-structured proforma containing demographic data, clinical features, and associated comorbidities was used to collect baseline data. A thorough cutaneous and systemic examination was done. Acne was graded for assessment of severity according to the global acne grading system. Clinical photographs according to standard global assessment was taken.
After an overnight fast (at least 12 h), venous blood samples were collected from the anterior cubital vein in all participants under complete aseptic conditions and analysed for fasting blood glucose (FBG) lipid profile, fasting insulin in the clinical biochemistry laboratory.
Operational definitions: Diagnosis of MetS was done on the basis of National Cholesterol Education Programme (NCEP) adult treatment panel III by the presence of 3 or more of the following criteria-
a) TGs value ≥ 150 mg/dl
b) HDL ≤ 40 mg/dl in males
c) FBS ≥ 110 mg/dl
d) Waist circumference of ≥ 102 cm
e) BP ≥ 130/85mmHg
Are the cut off points in Males[16].
A HOMA-IR value greater than 2.5 was predefined arbitrarily as insulin resistance. The HOMA-IR values were calculated using the following formula - Fasting Insulin (Microunits per Millilitre) × Fasting Glucose (mg/dl)/405.
Statistical procedures: Data were entered into MS Excel 2010. Data cleaning was carried out, and statistical analysis was carried out using SPSS software version 20.0.
Continuous variables such as. following normal distribution were presented as mean (standard deviation). Categorical variables were presented as frequency and percentage. Data were also tabulated and graphically represented.
The Student's t-test was used to compare continuous data. Additionally, the Pearson Chi-square test was used to compare categorical data.
Ethical considerations: The approval to conduct the study was sought from the Research monitoring committee and Institutional Ethics Committee (IEC) with the reference number: IEC/App/July/2022/014. Informed consent was obtained from the study participants before data collection. Informed consent was taken by the investigator, after explaining the purpose of the study. The freedom to withdraw from the study at any time during the interview was also explained prior to taking of the informed consent. Data were analyzed in aggregate and access to the collected data was limited only to me, my guide and co-guide.
Sample Size of Estimation: The sample size of for the study was determined using the formula
n= z2 pq/d2
n = required sample size
z = reliability coefficient (z score) of 1.96 at the 95% confidence level
p = Assuming the estimated prevalence of insulin resistance in the target population is 0.426 (42.6%) based on previous study 101
q = 1-p
d = allowable absolute error
Given the estimated prevalence of metabolic syndrome in the target population, the sample size calculation is as follows:
Assuming the estimated prevalence (p) is 0.426 (42.6%) based on previous studies, 101 and a margin of error (d) of 0.09 (9%):
n=(1.96)2×0.426×(1−0.426)/(0.09)2
n= 116.22
To account for potential non-response or incomplete data, the sample size was rounded up and adjusted to 126 participants.
Therefore, the total sample size calculated for this study was 126.
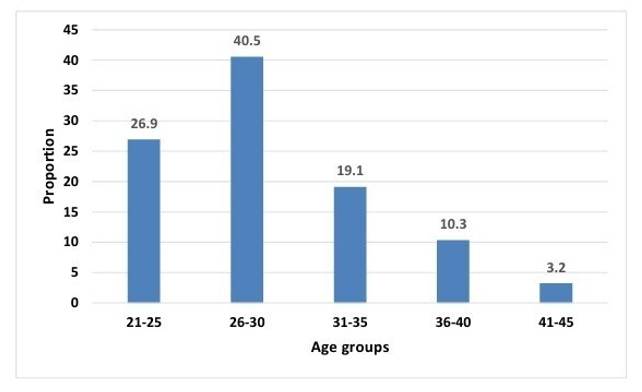
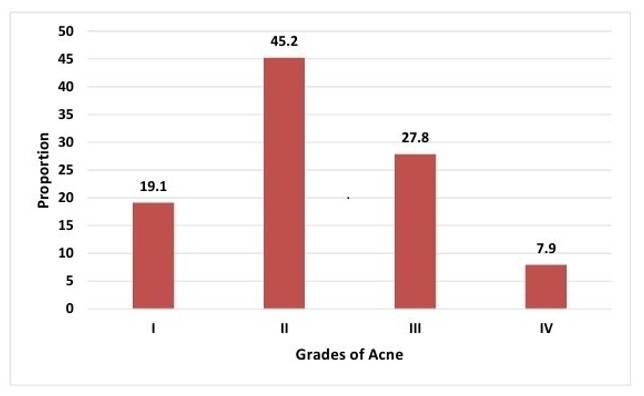
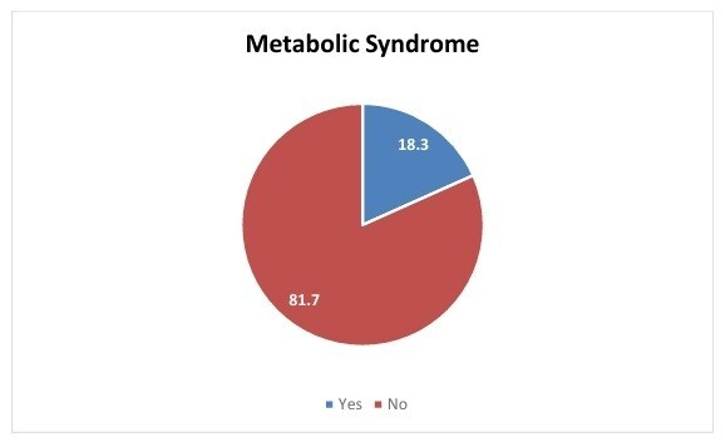
A total of 126 participants with acne vulgaris, and mean age of 29.1 years (SD=5.05) were enrolled: Age distribution displayed a predominance in the 26-30 age group (40.5%), followed by 21-25 (26.9%) and 31-35 (19.1%) age categories. Clinical parameters, including fasting blood sugar (92.5 mg/dl, SD=13.61), triglycerides (139.3 mg/dl, SD=43.63), and HDL (44.3 mg/dl, SD=8.57), fell within typical ranges. Acne grades were predominantly categorized as grade II (45.2%), followed by grades III (27.8%) and I (19.1%). Duration of acne varied, with 61.1% of participants reporting acne for ≤1 year and 34.9% reporting 1-5 years. Metabolic syndrome was diagnosed in 18.3% of participants according to NCEP-III criteria. Among individuals with metabolic syndrome, the highest prevalence was observed in the 26-30 age group (39.1%). More than half of participants with metabolic syndrome exhibited increased waist circumferences (≥102 cm, 56.5%) and elevated blood pressure (≥130/85 mm/Hg, 60.9%). HOMA IR ≥2.5 was reported in 32.5% of participants.
Among participants with raised HOMA IR, the 26-30 age group had the highest representation (34.1%). Acne grade II was most prevalent (36.6%) among individuals with raised HOMA IR. Acne duration of ≤1 year was predominant (63.4%) among participants with raised HOMA IR. The proportion of participants with raised fasting blood sugar was 17.4% among those with metabolic syndrome. Raised triglycerides were observed in 86.9% of individuals with metabolic syndrome. Elevated blood pressure (≥130/85 mm/Hg) was prevalent in 60.9% of participants with metabolic syndrome. HDL levels < 40 mg/dl were found in only 4.4% of individuals with metabolic syndrome. Waist circumference ≥102 cm was noted in 56.5% of participants with metabolic syndrome. Participants with raised HOMA IR had a higher proportion of individuals aged 21-25 years (29.3%). Among individuals with raised HOMA IR, 29.3% had acne grade III. Acne duration of 1-5 years was reported by 34.2% of participants with raised HOMA IR.
This study investigated the association between acne and metabolic syndrome, as well as insulin resistance, in post-adolescent male patients. It further assessed acne grade using the global acne grading system in patients diagnosed with metabolic syndrome and/or insulin resistance. Additionally, the study described the demographic profile of post adolescent male patients with acne.
The majority of participants were in their late twenties and early thirties, with 40.5% falling within the 26-30 age group and 26.9% in the 21-25 years group [Table. 1] [Fig. 2]. This indicates a substantial representation of post-adolescent and young adults experiencing acne, consistent with the growing recognition of "post-adolescent acne" as a distinct entity [Fig. 1].
|
Age Category (years) |
Frequency |
Proportion (%) |
|---|---|---|
|
21–25 |
34 |
26.9 |
|
26–30 |
51 |
40.5 |
|
31–35 |
24 |
19.1 |
|
36–40 |
13 |
10.3 |
|
41–45 |
4 |
3.2 |
|
Total |
126 |
100 |
Around 40% (n=51) belonged to 26-30 years of age, 26.9% (n=34) belonged to 21-25 years of age and 20% (n=24) were in 31-35 years of age group.
While mean fasting blood sugar fell within the normal range, indicating no impaired glucose metabolism, the high mean triglyceride (139.3±43.63 mg/dl) and low HDL (44.3±8.57 mg/dl) levels are features associated with metabolic syndrome and might link acne to systemic metabolic imbalances[17-19]. These findings raise the possibility of a link between acne and systemic metabolic imbalances. It is essential to recognize that the study's participants exhibited a normal fasting blood sugar range, indicating that impaired glucose metabolism may not be a contributing factor to acne in this cohort.
Anthropometric measurements provided valuable insights into the participants' physical characteristics. It reveals abdominal adiposity, as evidenced by the mean waist circumference of 87.7 cm, a known risk factor for insulin resistance and a component of metabolic syndrome. Additionally, the mean Basal metabolic rate (BMI) falling within the overweight range (25.6 kg/m2) suggests a potential association between higher BMI and acne[18]. This finding aligns with existing literature that associates higher BMI with skin conditions.
The study's comprehensive approach in assessing metabolic and anthropometric markers contributes significantly to understanding the multifaceted nature of the relationship between acne and metabolic syndrome. Compared to other Indian studies, our participants exhibited slightly higher mean triglyceride, HDL, BMI, systolic blood pressure (SBP), diastolic blood pressure (DBP) and waist circumference values[20, 21]. These differences suggest potential variations in the metabolic and anthropometric profiles of acne patients across different regional populations.
This study paints a clear picture of acne severity, revealing that nearly half (45.2%) of participants were diagnosed with Grade II acne [Table. 2] [Fig. 3]. This finding resonates with other Indian studies reporting similar predominance of Grade II acne among young men[22, 23]. Also, the majority (61.1%) reported experiencing acne for ≤1 year, indicating a potential trend toward seeking early medical attention. This positive shift towards early intervention may be attributed to increased awareness of acne's impact on mental health, the availability of effective treatments, and a focus on preventative care.
|
Grades of Acne |
Frequency |
Proportion (%) |
|---|---|---|
|
I |
24 |
19.1 |
|
II |
57 |
45.2 |
|
III |
35 |
27.8 |
|
IV |
10 |
7.9 |
|
Total |
126 |
100 |
Around 45% (n=57) belonged to grade II, 28% (n=35) belonged to grade III and 19% (n=24) belonged to grade I.
This study reported MetS in 18.3% of participants based on NCEP III criteria with a higher prevalence in the 26-30 years category (39.1%) [Table. 3] [Fig. 4]. This suggests a potential life stage susceptibility to MetS during young adulthood, possibly attributable to hormonal shifts or lifestyle alterations associated with this period. This observation poses a critical question regarding the pivotal role of MetS in the initiation or exacerbation of acne in this specific age group. The study reveals a noteworthy correlation between higher acne grades and the presence of MetS. This strengthens the hypothesis that metabolic imbalances characteristic of MetS, through shared inflammatory pathways or hormonal alterations, may contribute to acne severity. Further research is crucial to elucidate the underlying mechanisms driving this potential association.
|
Metabolic Syndrome |
Frequency |
Proportion (%) |
|---|---|---|
|
Yes |
23 |
18.3 |
|
No |
103 |
81.7 |
|
Total |
126 |
100 |
18.3% (n=23) study participants were found to be present with metabolic syndrome.
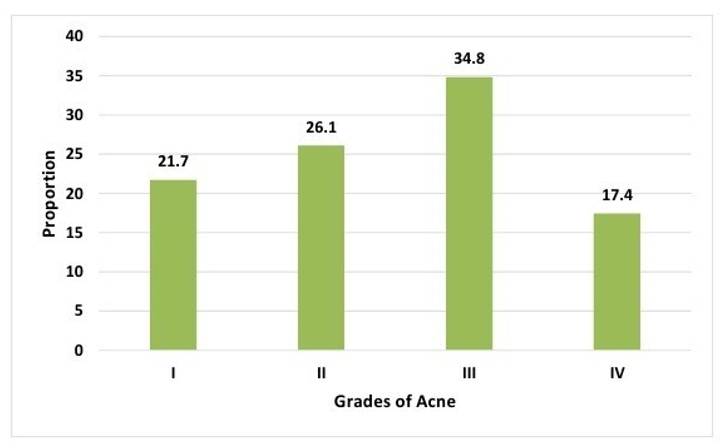
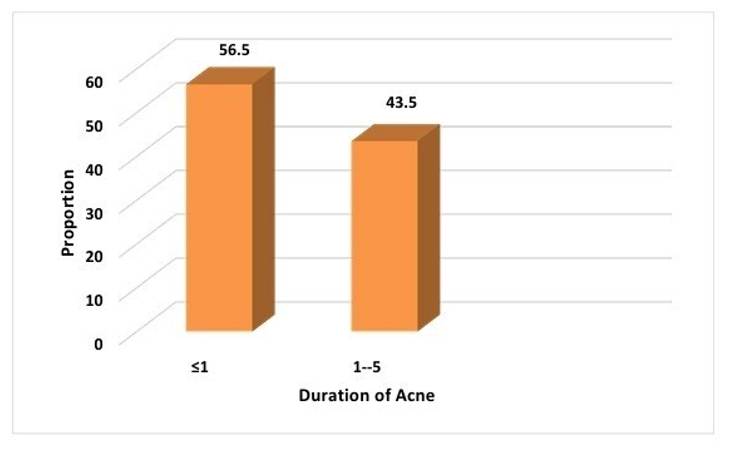
Similar Indian studies by Chandak S et al. and Nagpal et al. have reported comparable prevalence rates, supporting the consistency of these findings across diverse populations.[20][24] But a higher prevalence was reported by Podder I et al.[25] which suggest that geographical and demographic factors may contribute to variations in MetS prevalence. Furthermore, the study revealed that a majority of participants with MetS (56.5%) had acne for ≤1 year, emphasizing the early manifestation of MetS in the acne course [Table. 4] [Table. 5] [Fig. 5] [Fig. 6]. This highlights the importance of timely metabolic screening and intervention in acne patients.
|
Grades of acne |
Metabolic Syndrome |
|
|---|---|---|
|
Frequency |
Proportion |
|
|
I |
5 |
21.7 |
|
II |
6 |
26.1 |
|
III |
8 |
34.8 |
|
IV |
4 |
17.4 |
|
Total |
23 |
100 |
Around 34.8% (n=8) belonged to grade III, 26.1% (n=6) belonged to grade II and 21.7% (n=5) belonged to grade I.
|
Duration of acne (years) |
Metabolic Syndrome |
|
|---|---|---|
|
Frequency |
Proportion |
|
|
≤1 |
13 |
56.5 |
|
1-5 |
10 |
43.5 |
|
Total |
23 |
100 |
Of the total, 56.5% (n=13) had acne since ≤1 year and 43.5% (n=10) had acne from the last 1-5 years.
However, study unveils a fascinating paradox within the MetS subgroup. While 18.3% of participants met criteria for MetS, a notable dissociation emerged regarding lipid profiles. A majority (61.1%) maintained healthy HDL levels within the reference range. Conversely, a striking 86.9% exhibited elevated triglycerides, exceeding recommended thresholds. Conducting further research using advanced lipidomic and metabolomic approaches becomes imperative to elucidate the specific lipid species and metabolic pathways involved in this context. The study's results differ from those reported by Senkadhirdasan D et al. among Indian adults with MetS[26]. This discrepancy highlights the potential for geographical or demographic variations in metabolic patterns amongst individuals with co-existing acne and MetS. Investigating these variations through larger, multi-centre studies could broaden our understanding and inform region-specific management strategies.
Despite maintaining relatively healthy HDL levels, a notable proportion of individuals with MetS in the study exhibited markers of abdominal adiposity and elevated blood pressure, revealing hidden dimensions of metabolic imbalance.
More than half (56.5%) displayed increased waist circumference (waist circumference ≥102 cm), a risk factor for insulin resistance, a core component of MetS, and previously linked to acne severity. Additionally, a substantial 60.9% presented with elevated blood pressure (blood pressure ≥130/85 mm/Hg), emphasizing the need for a comprehensive approach to acne management in young men. Beyond focusing solely on visible acne lesions, this approach should incorporate strategies addressing metabolic comorbidities associated with MetS. Lifestyle modifications, early identification, and multidisciplinary collaboration are crucial components.
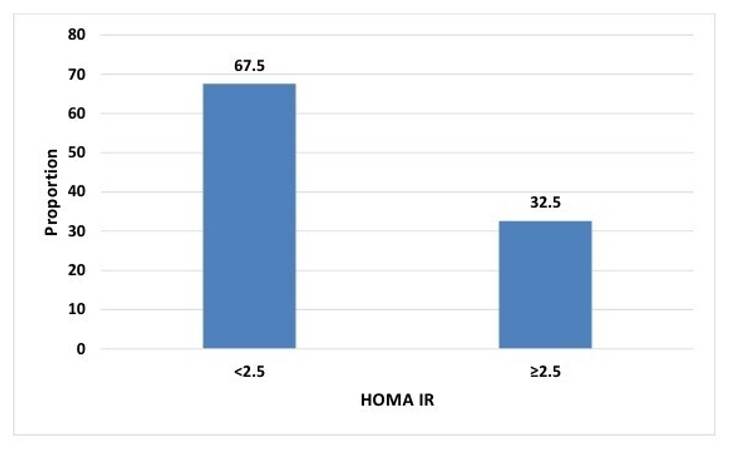
This study sheds further light on the role of insulin resistance in post-adolescent acne, revealing a concerning prevalence of 32.5% with HOMA-IR values exceeding 2.5 [Table. 6] [Fig. 7]. While the observed prevalence appears slightly higher than that reported in adult acne patients by Nagpal et al., the variations could stem from differences in age groups or study populations. Studies by Emiroglu N et al.[27] and Unluhizarci K et al.[28] have suggested that elevated insulin levels and subsequent insulin resistance contribute to increased androgen production, hyperkeratinisation of hair follicles, and sebum production, collectively promoting acne development.
| HOMA IR |
Frequency |
Proportion |
|---|---|---|
|
<2.5 |
85 |
67.5 |
|
≥2.5 |
41 |
32.5 |
|
Total |
126 |
100 |
32.5% (n=41) study participants had HOMA IR ≥2.5 and 67.5% (n=85) had HOMA IR <2.5. (HOMA-IR=Homeostatic Model Assessment of Insulin Resistance)
The implications for management are significant, emphasizing the need to consider metabolic factors in acne treatment for young men. Strategies aimed at improving insulin sensitivity through lifestyle modifications could offer dual benefits by addressing both acne severity and long-term metabolic health.
This study explores beyond the prevalence of insulin resistance (HOMA-IR) in post adolescent acne, unravelling its intricate relationships with age, severity, and duration. While 32.5% of participants exhibited elevated HOMA-IR, a concerning trend emerged within the 26-30 age group, showcasing a notably higher prevalence (34.1%). This suggests a potential peak in susceptibility to insulin resistance during this specific life stage, possibly due to hormonal fluctuations or lifestyle changes associated with young adulthood. This finding underscores the importance of research targeted towards this vulnerable age group experiencing persistent acne beyond adolescence.
Furthermore, the distribution of acne grades among those with raised HOMA IR highlighted a substantial proportion (36.6%) in grade II, indicating a moderate severity [Table. 7] [Fig. 8]. This observation is different from the findings of Soodan PS et al. where mean HOMA-IR was found more in grade IV acne among males,[29] suggesting a positive correlation between insulin resistance and acne severity. While further research is crucial to reconcile these discrepancies, it raises the possibility of nonlinear or age-dependent relationships between insulin resistance and acne severity.
|
Grades of acne |
Raised HOMA IR |
|
|---|---|---|
|
Frequency |
Proportion |
|
|
I |
9 |
21.9 |
|
II |
15 |
36.6 |
|
III |
12 |
29.3 |
|
IV |
5 |
12.2 |
|
Total |
41 |
100 |
Around 36.6% (n=15) belonged to grade II, 29.3% (n=12) belonged to grade III and 21.9% (n=9) belonged to grade I.
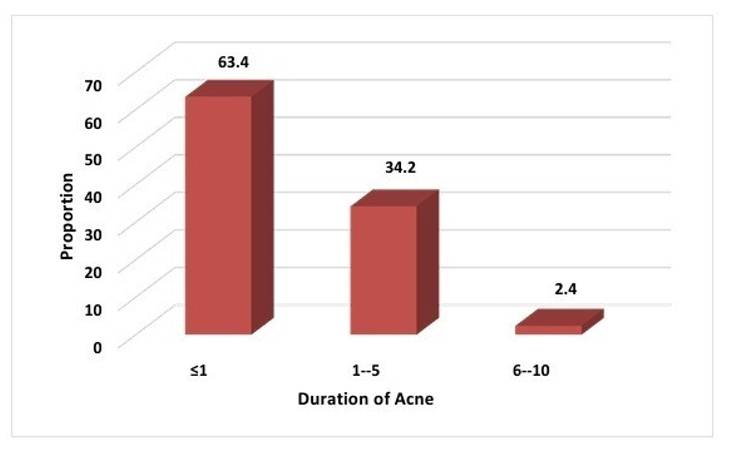
A noteworthy finding is that the majority of participants with elevated HOMA-IR (63.4%) had experienced acne for only one year or less [Table. 8] [Fig. 9]. This suggests that insulin resistance might manifest early in the course of acne development, even before visible symptoms become apparent. This emphasizes the importance of proactive metabolic screening and intervention in acne patients, potentially mitigating long-term health risks associated with insulin resistance and optimizing acne management outcomes.
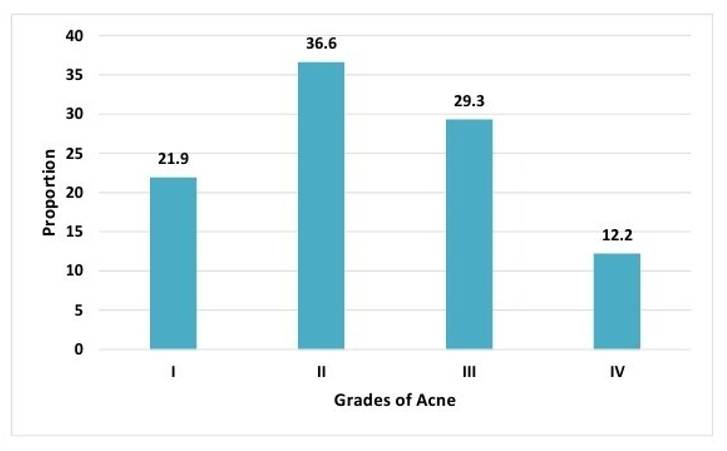
Table 8: Duration of acne distribution among study participants with raised HOMA IR (≥2.5)
|
Duration of acne (years) |
Raised HOMA IR |
|
|---|---|---|
|
Frequency |
Proportion |
|
|
≤1 |
26 |
63.4 |
|
1-5 |
14 |
34.2 |
|
6-10 |
1 |
2.4 |
|
Total |
41 |
100 |
Of total, 63.4% (n=26) had acne since ≤1 year, 34.2% (n=14) had from last 1-5 years and only 2.4% (n=1) had acne since 6-10 years.
This study sheds light on a potential link between MetS and acne, particularly its severity and early onset. Our findings suggest the importance of considering metabolic factors in acne management.
The cross-sectional design limits causal inference between metabolic factors and acne. The sample's size and demographic may not represent the broader population, especially since it predominantly includes males. Missing confounding variables like diet, genetics, and skincare may also restrict the study's ability to fully address acne's causes.
This study provides insights into the relationship between metabolic parameters and acne severity in post-adolescent males. It found an 18.3% prevalence of metabolic syndrome, higher in the 26-30 age group, and a significant correlation with elevated acne grades. Metabolic syndrome may appear early in acne development, highlighting the need for timely metabolic screening. The study also identifies unique metabolic patterns, particularly regarding HDL and triglycerides, warranting further research. These findings support a holistic approach to acne management, integrating metabolic screening and interventions targeting metabolic health to improve outcomes and enhance the understanding of acne's connection to metabolic health.
Funding: None
Conflict of Interest: No
1. The prevalence of acne in adults 20 years and older. Journal of the American Academy of Dermatology. 2008; 58 (1). Available from: https://doi.org/10.1016/j.jaad.2007.06.045
2. Can We Define Acne as a Chronic Disease?. American Journal of Clinical Dermatology. 2008; 9 (5). Available from: https://doi.org/10.2165/00128071-200809050-00001
3. Tumor necrosis factor-α −308 G/A and interleukin 10 −1082 A/G gene polymorphisms in patients with acne vulgaris. Journal of Dermatological Science. 2012; 68 (1). Available from: https://doi.org/10.1016/j.jdermsci.2012.07.001
4. Inflammation and Extracellular Matrix Degradation Mediated by Activated Transcription Factors Nuclear Factor-κB and Activator Protein-1 in Inflammatory Acne Lesions in Vivo. The American Journal of Pathology. 2005; 166 (6). Available from: https://doi.org/10.1016/s0002-9440(10)62479-0
5. Inflammatory Events Are Involved in Acne Lesion Initiation. Journal of Investigative Dermatology. 2003; 121 (1). Available from: https://doi.org/10.1046/j.1523-1747.2003.12321.x
6. Enzymes involved in the biosynthesis of leukotriene B4 and prostaglandin E2 are active in sebaceous glands. Journal of Molecular Medicine. 2006; 84 (1). Available from: https://doi.org/10.1007/s00109-005-0715-8
7. Zileuton prevents the activation of the leukotriene pathway and reduces sebaceous lipogenesis. Experimental Dermatology. 2010; 19 (2). Available from: https://doi.org/10.1111/j.1600-0625.2009.00929.x
8. Harvey A, Huynh TT. Inflammation and acne: Putting the pieces together. Journal of Drugs in Dermatology. 2014;13(4):459–63.
9. What's new in the physiopathology of acne?. British Journal of Dermatology. 2015; 172 (S1). Available from: https://doi.org/10.1111/bjd.13634
10. Ogé LK, Broussard A, Marshall MD. Acne Vulgaris: Diagnosis and Treatment. 2019 p. 475–84.
11. A comparative study of biological and metabolic biomarkers between healthy individuals and patients with acne vulgaris. Medicine. 2017; 96 (45). Available from: https://doi.org/10.1097/md.0000000000008554
12. Acne Vulgaris: Pathogenesis, Treatment, and Needs Assessment. Dermatologic Clinics. 2012; 30 (1). Available from: https://doi.org/10.1016/j.det.2011.09.001
13. Insulin‐like growth factor‐I gene polymorphism in acne vulgaris. Journal of the European Academy of Dermatology and Venereology. 2013; 27 (2). Available from: https://doi.org/10.1111/j.1468-3083.2011.04299.x
14. Correlation Between Serum Levels of Insulin-like Growth Factor 1, Dehydroepiandrosterone Sulfate, and Dihydrotestosterone and Acne Lesion Counts in Adult Women. Archives of Dermatology. 2005; 141 (3). Available from: https://doi.org/10.1001/archderm.141.3.333
15. Regulation of Human Sebaceous Glands. Journal of Investigative Dermatology. 2004; 123 (1). Available from: https://doi.org/10.1111/j.1523-1747.2004.t01-2-.x
16. NCEP-Defined Metabolic Syndrome, Diabetes, and Prevalence of Coronary Heart Disease Among NHANES III Participants Age 50 Years and Older. Diabetes. 2003; 52 (5). Available from: https://doi.org/10.2337/diabetes.52.5.1210
17. Why Syndrome X? From Harold Himsworth to the Insulin Resistance Syndrome. Cell Metabolism. 2005; 1 (1). Available from: https://doi.org/10.1016/j.cmet.2004.12.001
18. Pathophysiology of the Metabolic Syndrome: Implications for the Cardiometabolic Risks Associated With Type 2 Diabetes. The American Journal of the Medical Sciences. 2012; 343 (1). Available from: https://doi.org/10.1097/maj.0b013e31823ea214
19. Metabolic syndrome and skin: Psoriasis and beyond. Indian Journal of Dermatology. 2013; 58 (4). Available from: https://doi.org/10.4103/0019-5154.113950
20. Harmonizing the Metabolic Syndrome. Circulation. 2009; 120 (16). Available from: https://doi.org/10.1161/circulationaha.109.192644
21. Acne Vulgaris. Encyclopedia of Pharmacy Practice and Clinical Pharmacy. 2019; Available from: https://doi.org/10.1016/b978-0-12-812735-3.00552-5
22. Dermatology: how to manage acne vulgaris. Drugs in Context. 2021; 10 Available from: https://doi.org/10.7573/dic.2021-8-6
23. The age-old problem of acne. International Journal of Women's Dermatology. 2017; 3 (2). Available from: https://doi.org/10.1016/j.ijwd.2016.11.002
24. Why do humans get acne? A hypothesis. Medical Hypotheses. 2020; 134 Available from: https://doi.org/10.1016/j.mehy.2019.109412
25. Evaluation of Psychological Wellbeing and Social Impact of Combined Facial and Truncal Acne: a Multi-national, Mixed-Methods Study. Dermatology and Therapy. 2022; 12 (8). Available from: https://doi.org/10.1007/s13555-022-00768-0
26. Hoover E, Aslam S, Krishnamurthy K. Physiology, Sebaceous Glands. StatPearls. 2022 Oct.
27. The skin function: a factor of anti-metabolic syndrome. Diabetology & Metabolic Syndrome. 2012; 4 (1). Available from: https://doi.org/10.1186/1758-5996-4-15
28. Suva MA, Patel AM, Sharma N, Bhattacharya C, Mangi RK. A Brief Review on Acne Vulgaris: Pathogenesis, Diagnosis and Treatment. STM Journals. 2014;4(3):1–12.
29. Acne vulgaris. Nature Reviews Disease Primers. 2015; 1 (1). Available from: https://doi.org/10.1038/nrdp.2015.29
Subscribe now for latest articles and news.