

Journal of Medical Sciences and Health
DOI: 10.46347/jmsh.v12.i1.25.186
Year: 2026, Volume: 12, Issue: 1, Pages: 115-119
Case Report
Gauri Kale 1, Tulsi Chhadi 2, Kirti Jaiswal 3, Swati Gadhave 4, Satish Helwatkar 2
1Junior Resident, Department of Pathology, Government Medical College, Nagpur - 440003, Maharashtra, India.
2Associate Professor, Department of Pathology, Government Medical College, Nagpur - 440003, Maharashtra, India.
3Professor and Head, Department of Pathology, Government Medical College, Nagpur - 440003, Maharashtra, India.
4Assistant Professor, Department of Pathology, Government Medical College, Nagpur - 440003, Maharashtra, India. Address for correspondence: Swati Gadhave, Assistant Professor, Department of Pathology, Government Medical College, Nagpur - 440003, Maharashtra, India.
E-mail: [email protected]
Received Date:17 May 2025, Accepted Date:29 August 2025, Published Date:11 May 2026
Juvenile papillomatosis (JP) of the breast is a rare benign proliferative lesion predominantly affecting young women. Although benign, JP is clinically significant due to its potential association with an increased risk of breast cancer, particularly in patients with a family history of malignancy. A 19-year-old female presented with a firm, mobile lump in the right breast, classified as BIRADS-IV on ultrasonography. Fine needle aspiration cytology (FNAC) suggested a papillary lesion and histopathological examination after surgical excision confirmed JP. The specimen revealed cystically dilated ducts, papillary projections, apocrine metaplasia, and sclerosing adenosis with immunohistochemistry (IHC) positive for CK 5/6 confirming the benign nature of the lesion. JP poses diagnostic challenges due to its clinical and radiological overlap with both benign and malignant breast lesions. Histopathology remains the gold standard for diagnosis, often revealing characteristic features such as ductal papillomatosis and intraluminal secretions. The association between JP and breast cancer risk, particularly in patients with a family history, underscores the need for long-term surveillance. Surgical excision is the standard treatment to confirm diagnosis and prevent recurrence. This case highlights the diagnostic complexity of JP and reinforces the importance of histopathological confirmation and complete surgical excision. Long-term follow-up is essential, especially for patients with a family history of breast cancer. Further research is needed to elucidate the genetic basis of JP and its potential link to breast cancer risk.
Juvenile papillomatosis (JP), commonly referred to as Swiss cheese disease, is a rare benign breast condition that primarily affects young women[1]. It often presents similarly to fibroadenoma, appearing as a localized, multinodular mass during breast examinations[2]. However, JP lesions tend to be firmer and larger than fibroadenomas and may sometimes be tender. According to literature, these tumors are most frequently found in the upper and outer quadrant of the breast[3]. Since its initial description in 1980, several studies have suggested a link between JP and a family history of breast cancer[4].
Reports indicate that 26-58% of JP patients have a first/second degree relative with a history of breast cancer. Additionally, recurrent bilateral JP combined with a positive family history has been recognized as a risk factor for invasive breast cancer[5]. Histologically, JP may exhibit epithelial atypia, though no direct correlation has been established between the grade of atypia and the subsequent development of breast cancer[6].
A 19-year-old female presented with a non-painful, firm, mobile lump in her right breast, located in the upper outer quadrant, measuring 3 x 2 cm. The overlying skin and nipple-areola complex appeared normal. Both axillae and the opposite breast were unremarkable. No significant family history of breast carcinoma noted. An ultrasound showed a solid lesion with a cystic component measuring 2.5x1.6cm at the 10 o’ clock position, classified as BIRADS-IV, indicating a suspicious lesion. Fine needle aspiration cytology (FNAC) revealed a blood-mixed serous aspirate with smears showing benign epithelial cells, branching papillae, clusters of epithelial
cells, dispersed apocrine cells, foamy cells and a few bipolar nuclei, all in a proteinaceous background, suggesting a papillary lesion [Fig. 1].
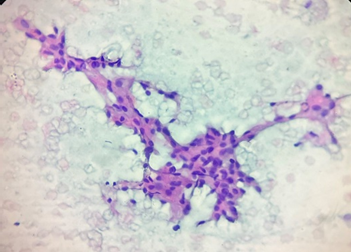
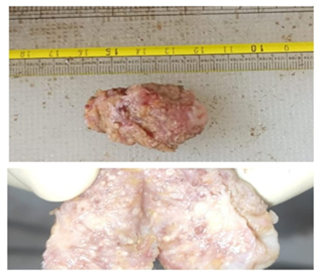
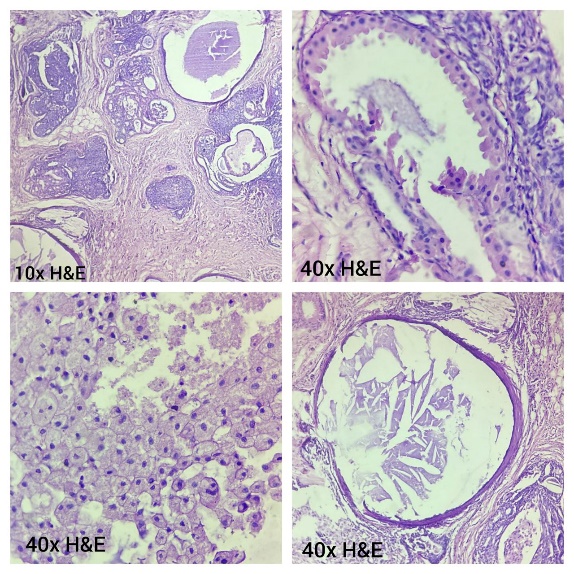
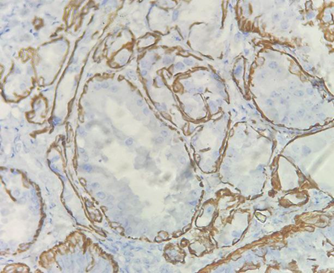
Surgical excision of the lump resulted in a tissue specimen measuring 4 x 2.5 x 1.5 cm with multiple cystic spaces filled with a cheese-like material on gross examination [Fig. 2]. Histopathology revealed multiple hyperplastic, cystically dilated ducts with papillary projections, lined by luminal and myoepithelial cells. Other findings included intraluminal eosinophilic secretion, foamy macrophages, cholesterol clefts, foci of sclerosing adenosis, apocrine metaplasia and areas of usual ductal hyperplasia [Fig. 3]. These features were consistent with juvenile papillomatosis. Immunohistochemistry (IHC) positivity for CK 5/6 highlighted the myoepithelial cells, confirming the benign nature of the lesion [Fig. 4].
JP is an uncommon histopathological condition, typically affecting women under the age of 30. While considered benign, JP is significant due to its potential association with an increased risk of breast cancer, particularly in patients with a strong family history of malignancy. Its clinical and radiological overlap with other benign and malignant breast lesions makes accurate diagnosis challenging. The present case of a 19-year-old female with a firm, mobile breast lump classified as BIRADS-IV on ultrasonography and later confirmed as JP histopathologically aligns with various reported cases in the literature, underscoring the diagnostic difficulty and management challenges associated with JP.
| Study | Age / Sex | Complaints | Radiology | FNAC | Biopsy | Size (cm) | Gross Findings | Microscopy | IHC – CK 5/6 |
|---|---|---|---|---|---|---|---|---|---|
| Present study | 19/F | Non-tender, mobile lump | USG – BIRADS 4 | Clusters of epithelial cells, branching papillae | – | 4 × 2.5 × 1.5 | Cystic spaces filled with cheese-like material | Multiple hyperplastic, cystically dilated ducts, apocrine metaplasia, foamy macrophages, areas of UDH | Positive |
| Hameed R et al. [13], 2023 | 38/F | Lump without pain | USG – solid cystic lesion | Mucinous carcinoma | – | 4 × 3 × 2 | Swiss cheese appearance | Ducts lined by bilayer epithelium showing hyperplasia | – |
| Al-Qurashi et al. [11], 2024 | 13/F | Cyclical pain with discharge | USG – BIRADS 4 | – | – | – | – | Multiple papillomatosis | – |
| Kafadar MT et al. [16], 2017 | 24/F | No pain, discharge | USG – BIRADS 4b | – | – | 3 × 2 | – | Cysts lined by both epithelial layers | Positive |
| Kafadar MT et al. [16], 2017 | 11/F | Lump without pain | USG – solid lesion | – | Fibroglandular lesion | 7 × 6 | – | – | – |
| Lad S et al. [15], 2014 | 16/F | Non-tender lump | USG – lobulated heterogeneous lesion with cystic spaces | – | Florid ductal hyperplasia, apocrine metaplasia, microcysts | 3 | Numerous cysts | Extensive proliferative fibrocystic changes, multiple cystic spaces | – |
| Wang et al. [9], 2014 | 11/F | Tender mass | USG – irregularly spaced, inhomogeneous lesion | – | – | 3 × 3 × 2 | – | Multiple dilated ducts, intracystic papillary epithelial proliferation with apocrine metaplasia | – |
| Gunes et al. [8], 2024 | 37/F | Discharge from nipple | MRI – millimetric cyst | – | Ductal hyperplasia, sclerosing adenosis | – | – | – | – |
| Zavieh et al. [14], 2020 | 14/F | Lump with nipple discharge | USG – BIRADS 4b | Intraductal papilloma without atypia | – | 3 × 1.2 × 1.8 | – | Florid papillary hyperplasia | – |
JP typically presents as a solitary, palpable breast mass, often firm, mobile and painless, similar to fibroadenomas. However, variations exist, including bilateral involvement, multifocality and even associated nipple discharge. Imaging modalities such as ultrasound, mammography and MRI often reveal heterogeneous masses with cystic areas, raising suspicion for malignancy, as seen in our case.
Vandeweerd et al. [7] emphasized that JP can mimic malignancies due to its heterogeneous, cystic and sometimes calcified appearance on imaging. In their cases, MRI revealed microcalcifications and fibrocystic distortions, reinforcing the necessity for histopathological confirmation. Similarly, Güneş et al. [8] reported a case where MRI showed ductal dilatation and heterogeneous enhancement, classified as BIRADS 4b,
raising suspicion of malignancy despite benign core biopsy findings. Wang et al. [9] documented bifocal JP in a premenarchal female, highlighting the rare multifocal nature of JP in paediatric populations. In our case, ultrasonography identified a solid-cystic lesion with concerning features (BIRADS-IV), necessitating histological evaluation to rule out malignancy.
The association between JP and increased breast cancer risk remains debated, but a positive family history is frequently reported in JP patients. Rosen et al. [1] noted that up to 50% of JP patients have a family history of breast cancer. Bazzocchi et al. [10] similarly documented familial clustering, with one case presenting a mother-daughter dyad, where the mother developed ductal carcinoma in situ (DCIS). Al-Qurashi et al. [11] reported a 13-year-old JP patient with a strong family history of breast cancer, underscoring the importance of genetic counselling and long-term monitoring.
Despite these associations, BRCA1/2 mutations are not commonly identified in JP patients. Vandeweerd et al. [7] reported a BRCA2-negative patient with JP, suggesting that other genetic factors may contribute to its pathogenesis. Future studies utilizing next-generation sequencing could help uncover additional genetic mutations implicated in JP.
Histopathological examination remains the gold standard for JP diagnosis, demonstrating characteristic features such as ductal papillomatosis, cystically dilated ducts with papillary projections, intraluminal eosinophilic secretions, foamy macrophages, cholesterol clefts and usual ductal hyperplasia. Our case exhibited these hallmark features with additional findings of apocrine metaplasia and sclerosing adenosis.
Rosen et al. [12] (1980) first described JP as a benign entity with proliferative changes, reinforcing its distinct histological features. Bazzocchi et al. [10] confirmed these findings in their series of 13 patients, noting a high frequency of sclerosing adenosis and apocrine changes. In the report by Hameed et al. [13], JP was initially misdiagnosed as mucinous carcinoma based on FNAC, further demonstrating the diagnostic challenge and the indispensable role of histopathology in differentiating JP from malignant lesions. Mohammadi Zavieh et al. [14] described florid papillary hyperplasia in a 14-year-old, where core needle biopsy (CNB) was instrumental in guiding management, though surgical excision was ultimately required for definitive diagnosis.
In some cases, JP may harbour atypical ductal hyperplasia or even ductal carcinoma in situ (DCIS), warranting careful histopathological evaluation. Sedloev et al. [6] reported JP coexisting with micropapillary DCIS in a 15-year-old female, highlighting the potential for malignant transformation. Similarly, Bazzocchi et al. [10] identified DCIS in two of their JP cases, emphasizing the need for long-term surveillance. In our case, IHC for CK 5/6 confirmed the presence of myoepithelial cells, ensuring the benign nature of the lesion.
Surgical excision is the standard treatment for JP to confirm diagnosis and prevent recurrence. In our case, complete excision was performed and histopathology confirmed JP.
Lad et al. [15] emphasized the importance of wide local excision, particularly in young patients, to minimize recurrence while preserving breast tissue. Similarly, Kafadar et al. [16] advocated conservative surgical approaches in paediatric patients to maintain favourable cosmetic outcomes. Patterson et al. [17] highlighted the risk of recurrence following incomplete excision, reinforcing the need for clear margins during surgery. In cases with high-risk histological features (e.g. atypical ductal hyperplasia or DCIS), mastectomy may be considered, though its necessity remains controversial.
Long-term surveillance is crucial for JP patients, especially those with a family history of breast cancer. Regular follow-up with clinical breast examination and ultrasonography is recommended, as emphasized by Al-Qurashi et al. [11]. In cases with associated atypia or strong familial risk, MRI screening may be beneficial. Study done by Peter Rosan et al. [18] demonstrated a 10% incidence of subsequent breast carcinoma in patients of juvenile papillomatosis.
In Tan TY et al. [19] report a rare association between JP and neurofibromatosis type 1 (NF1) in male infants, particularly those with café-au-lait spots, suggesting a potential genetic link. Their findings highlight the molecular interplay between NF1 and Noonan syndrome (NS), implicating shared pathways in cell proliferation and tumorigenesis. While our case illustrates the classic, isolated nature of JP in young females Tan TY et al.’s [19] work identifies a unique demographic — male infants with NF1 — where JP may emerge as a rare manifestation of genetic predisposition. Together, these findings underscore the importance of histopathological confirmation and genetic evaluation in atypical cases, calling for further research into the molecular underlying JP and its potential ties to syndromes like NF1.
Our case aligns with existing literature, reaffirming that JP remains a challenging diagnosis due to its overlapping clinical, radiological and cytological features with other benign and malignant breast lesions. Histopathology is essential for definitive diagnosis and complete surgical excision is the preferred treatment to prevent recurrence. The potential link between JP and future breast cancer risk, particularly in patients with a family history, underscores the importance of long-term follow-up. Further research is needed to elucidate the genetic basis of JP and optimize management strategies for this rare but clinically significant entity.
1. Juvenile papillomatosis of the breast and family history of breast carcinoma. Cancer. 1982; 49 (12). Available from: https://doi.org/10.1002/1097-0142(19820615)49:12<2591::aid-cncr2820491230>3.0.co;2-o
2. Juvenile Papillomatosis (JP) of the Female Breast (Swiss Cheese Disease) - Role of Breast Ultrasonography. Ultraschall in der Medizin - European Journal of Ultrasound. 2005; 26 (01). Available from: https://doi.org/10.1055/s-2004-813122
3. Mammary juvenile papillomatosis (“Swiss cheese” disease): Study of 121 cases reiterates need for long-term follow-up. The Breast Journal. 2018; 24 (6). Available from: https://doi.org/10.1111/tbj.13092
4. Juvenile papillomatosis of the breast in a male infant with Noonan Syndrome, café au lait spots, and family history of breast carcinoma. Pediatric Blood & Cancer. 2005; 45 (7). Available from: https://doi.org/10.1002/pbc.20323
5. Juvenile Papillomatosis (Swiss-Cheese Disease) of Breast in an Adult Male With Sequential Diagnoses of Ipsilateral Intraductal, Invasive, and Widely Metastatic Carcinoma: A Case Report and Review of the Disease in Males. International Journal of Surgical Pathology. 2017; 25 (6). Available from: https://doi.org/10.1177/1066896917705198
6. Sedloev T, Bassarova A, Angelov K, Vasileva M, Asenov Y. Combination of juvenile papillomatosis, juvenile fibroadenoma and intraductal carcinoma of the breast in a 15-Year-old girl. <I>Anticancer Research</I> 2015;35:5027-5029.
7. Juvenile Papillomatosis: A Case Report. European Journal of Breast Health. 2019; 15 (2). Available from: https://doi.org/10.5152/ejbh.2019.4362
8. Juvenile papillomatosis: A case report. Turkish Journal of Surgery. 2024; 40 (2). Available from: https://doi.org/10.47717/turkjsurg.2022.4745
9. Bifocal juvenile papillomatosis as a marker of breast cancer: A case report and review of the literature. Oncology Letters. 2014; 8 (6). Available from: https://doi.org/10.3892/ol.2014.2600
10. Juvenile Papillomatosis (Epitheliosis) of the Breast: A Clinical and Pathologic Study of 13 Cases. American Journal of Clinical Pathology. 1986; 86 (6). Available from: https://doi.org/10.1093/ajcp/86.6.745
11. Juvenile Papillomatosis of the Breast: a Report of Two Cases with Review of the literature. Medical Archives. 2024; 78 (2). Available from: https://doi.org/10.5455/medarh.2024.78.170-173
12. Juvenile papillomatosis (Swiss cheese disease) of the breast. The American Journal of Surgical Pathology. 1980; 4 (1). Available from: https://doi.org/10.1097/00000478-198002000-00001
13. Swiss cheese disease: A rare case of juvenile papillomatosis of the breast mimicking carcinoma in a young female. Indian Journal of Pathology and Microbiology. 2023; 66 (4). Available from: https://doi.org/10.4103/ijpm.ijpm_597_21
14. Papillary Lesion of the Breast in a Young Girl Suspicious to Juvenile Papillomatosis, Clinical Decision Making in a Multi-disciplinary Team and Review of the Literature. Archives of Breast Cancer. 2020; 7 (3). Available from: https://doi.org/10.32768/abc.202073100-103
15. Juvenile Papillomatosis: A Case Report and Literature Review. Clinical Breast Cancer. 2014; 14 (5). Available from: https://doi.org/10.1016/j.clbc.2014.03.003
16. Juvenile Papillomatosis of the Breast in a Pre-Pubertal Girl: An Uncommon Diagnosis. European Journal of Breast Health. 2018; 14 (1). Available from: https://doi.org/10.5152/ejbh.2017.3828
17. A Case of Juvenile Papillomatosis, Aka “Swiss Cheese Disease”. The Breast Journal. 2013; 19 (4). Available from: https://doi.org/10.1111/tbj.12137
18. Juvenile Papillomatosis of the Breast: A Follow-Up Study of 41 Patients Having Biopsies Before 1979. American Journal of Clinical Pathology. 1990; 93 (5). Available from: https://doi.org/10.1093/ajcp/93.5.599
19. Juvenile papillomatosis of the breast associated with neurofibromatosis 1. Pediatric Blood & Cancer. 2007; 49 (3). Available from: https://doi.org/10.1002/pbc.20956
Subscribe now for latest articles and news.